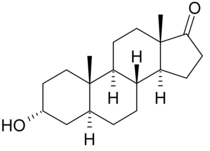
Androsterone
 |
|
 |
|
| Clinical data | |
|---|---|
| ATC code |
|
| Identifiers | |
|
|
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| ECHA InfoCard | 100.000.159 |
| Chemical and physical data | |
| Formula | C19H30O2 |
| Molar mass | 290.440 g/mol |
| 3D model (Jmol) | |
|
|
|
|
Androsterone, or 5α-androstan-3α-ol-17-one, is an endogenous steroid hormone, neurosteroid, and putative pheromone. It is a weak androgen with a potency that is approximately 1/7th that of testosterone. Androsterone is a metabolite of testosterone and dihydrotestosterone (DHT). In addition, it can be converted back into DHT via 3α-hydroxysteroid dehydrogenase and 17β-hydroxysteroid dehydrogenase, bypassing conventional intermediates such as androstenedione and testosterone, and as such, can be considered to be a metabolic intermediate in its own right. Androsterone is also known to be an inhibitory androstane neurosteroid, acting as a positive allosteric modulator of the GABAA receptor, and possesses anticonvulsant effects. The unnatural enantiomer of androsterone is more potent as a positive allosteric modulator of GABAA receptors and as an anticonvulsant than the natural form. Androsterone's 3β-isomer is epiandrosterone, and its 5β-epimer is etiocholanolone. The 3β,5β-isomer is epietiocholanolone.
...
Wikipedia
