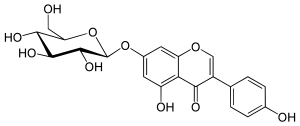
Genistin
 |
|
| Names | |
|---|---|
|
IUPAC name
5-hydroxy-3-(4-hydroxyphenyl)-7-[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxychromen-4-one
|
|
| Other names
Genistoside
Genistine Genistein 7-glucoside Genistein glucoside Genistein-7-glucoside Genisteol 7-monoglucoside Glucosyl-7-genistein Genistein 7-O-beta-D-glucoside |
|
| Identifiers | |
|
3D model (Jmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.120.406 |
|
PubChem CID
|
|
|
|
|
|
| Properties | |
| C21H20O10 | |
| Molar mass | 432.37 g/mol |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
|
|
|
|
|
| Infobox references | |
Genistin is an isoflavone found in a number of dietary plants like soy and kudzu. It was first isolated in 1931 from the 90% methanol extract of a soybean meal, when it was found that hydrolysis with hydrochloric acid produced 1 mole each of genistein and glucose. Chemically it is the 7-O-beta-D-glucoside form of genistein and is the predominant form of the isoflavone naturally occurring in plants. In fact Studies in the 1970s revealed that 99% of the isoflavonoid compounds in soy are present as their glucosides. The glucosides are converted by digestive enzymes in the digestive system to exert their biological effects. Genistin is also converted to a more familiar genistein, thus, the biological activities including antiatherosclerotic, estrogenic and anticancer effects are analogous.
When ingested along the diet, genistin is readily converted to its aglycone form, genistein. It is hydrolyzed by removing the covalently bound glucose to form genistein and that genistein is the form of the compound that is absorbed in the intestine and is the form responsible for the biological activities of the isoflavone. The digestive metabolism was first demonstrated in 2002 that the gut microflora played a large role in the conversion of genistin to genistein. It was later found that enzymes present in the human small intestine and liver also have the ability to convert the isoflavone. Hydrolysis actually starts very quickly in the digestive system once genistin is ingested, conversion begins in the mouth and then continues in the small intestine. Moreover, both human saliva and the intestinal cell-free extract from mice can cause the complete conversion.
...
Wikipedia
